PPE Certification Program
Non-Powered Air Purifying Respirators

During the COVID-19 global pandemic, as the world sees an increase in the demand for Personal Protective Equipment (PPE), and specifically for face masks, third-party approval of protective equipment is critical to ensure that it meets the required safety specifications, before being placed on the market. LC compliance reports provide manufacturers of respirators with the validation that their product provides the minimum level of protection against the intended risk.
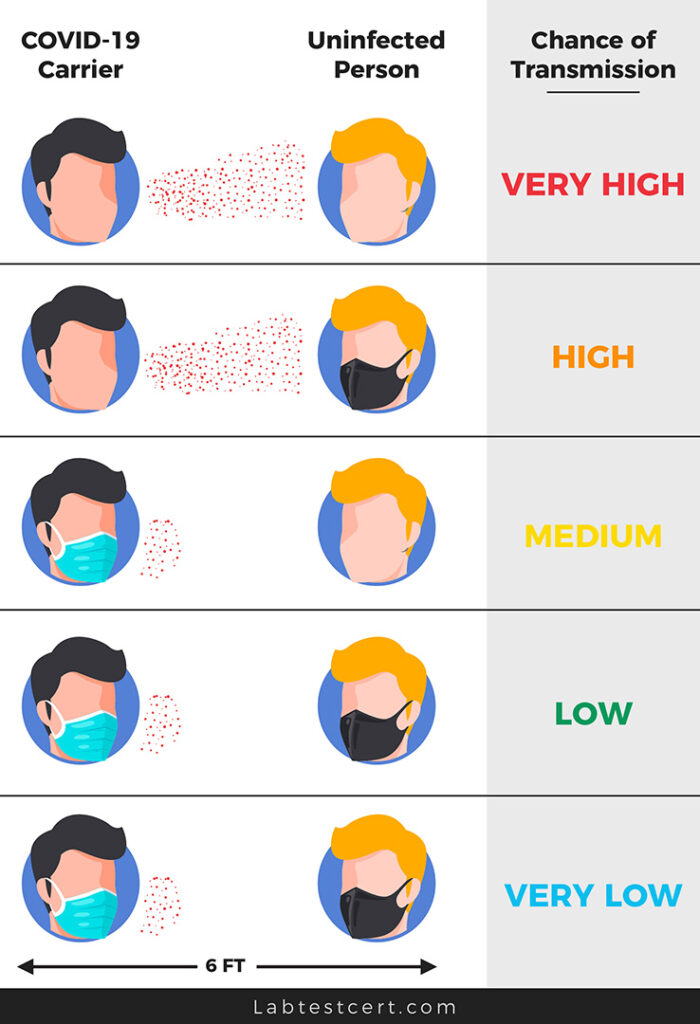
There are different types of protective masks available on the market, such as reusable (fabric) masks, surgical masks, and respirators. The filtration efficiency varies between masks based on many different factors.
What Is A Respirator?
As the Coronavirus outbreak continues, we have all become familiar with protective face masks, especially the N95 respirators. Compared with other types of protective masks, N95 respirators are designed to form a seal around the user’s nose and mouth, which may not be the case with other types masks, like for example, surgical masks. N95 respirators filtrate most particles larger than 0.06 microns in diameter and therefore, protect from viruses like the coronavirus, which usually has a diameter above 0.08 microns.

What does N95 stand for?
This is the rating of a non-powered filtering respirator that’s “not resistant to oil” and that has a minimum efficiency rating of 95%. What does that mean? This means that less than 5% of particles pass through the mask when tested using sodium chloride aerosol.
There are 3 main types of (non-powered) Air-Purifying Respirators:
N (Non-resistant to oil)
R (Resistant to oil)
P (oil Proof)
| N95 | KN95 | FFP2 | P2 | Korea 1st Class | DS | |
|---|---|---|---|---|---|---|
| Where | 
US & Canada |

China |

Europe |

Australia & New Zealand |

South Korea |

Japan |
|
Specification
|
NIOSH-42CFR84
|
GB2626-2006
|
EN 149:2001+ A1:2009
|
AS/NZ 1716:2012
|
KMOEL 2017-64
|
JMHLW-Notification 214, 2018 |
Having personal protective equipment tested by a third-party ensures that products such as Air-Purifying Respirators meet the requirements of the applicable product standards of the country in which they will be used and therefore, that it will not cause harm to the users. As each country’s regulations, and consequently each standard, are different, it is important to inform the third-party agency of which market the product is intended for, when submitting it for evaluation.
For respirators without an exhalation valve, two of the main tests required are: The Airflow Resistance Test and the Particulate Filtration Efficiency (PFE).
The Airflow Resistance test consists of determining the resistance of the mask to inhalation and exhalation. For each determination, the mask is mounted on the test equipment and the air is set to flow at a rate of 85 liters per minute. Respirators must meet the following requirements:


- 20 samples must be tested
- For 25 hours prior to the test, samples are conditioned at 85% relative humidity and 38°C
- For N-series respirators, a stream of solid sodium chloride particulate aerosol is created with tight constraints on the particle size distribution
- The aerosol is neutralized and is conditioned to a temperature of 25°C and a relative humidity of 30%.
- Single filter respirators are tested at a continuous airflow rate of 85 litres per minute
- The efficiency of the filter shall be monitored and recorded throughout the test period by measuring the particle concentration before and after the test sample.
- The test continues until the minimum efficiency is achieved or until at least 200±5 mg has contacted the filter

- Masks are classified based on the fraction of sodium chloride that passes through. N95 masks must filter out at least 95% of the NaCl solution, N99 masks filter at least 99% and N100 masks filter at least 99.97%
- The efficiency measured for each of the 20 samples must be equal to or greater than the filter efficiency criterion (e.g. 95% for N95).




Get In Touch